 +565 975 658
+565 975 658
 info@premiumcoding.com
info@premiumcoding.com
 Monday - Friday, 8.00 - 20.00
Monday - Friday, 8.00 - 20.00
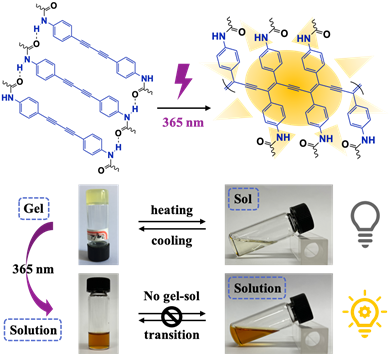
Polydiacetylenes (PDAs) are usually fabricated through topochemical polymerization initiated remotely by heat or UV irradiation without any additives. It has been widely acknowledged that the pre-arrangement of monomers is crucial for the 1, 4-addition topochemical transformation. So far, much effort has been devoted to employing supramolecular interactions to align diacetylenes into such ordered systems as Langmuir-Blodgett film, crystals, liquid crystals, monolayers, gels, self-assemblies and so forth. However, most of these strategies were generally conducted by heating or high energy irradiation, like 254-nm light or even γ-ray, which may bring irreversible damage to material itself and integrated devices. Thus, a mild condition with lower energy irradiation is urgently required. Moreover, due to the well-defined alternating ‘ene-yne’ conjugated skeleton, PDAs have shown broad application prospects in chemo/bio sensors, photocatalysis, cell imaging, 3D printing, photo patterning, anticounterfeiting and encryption, organic optoelectronic materials, etc. Therefore, using 365-nm light to initiate the topochemical polymerization without employment of any additional reagents is appealing but remains a great challenge.
Organogel systems are considerably favorable for the pre-arrangement of monomers for topochemical transformation and polymerization. Inspired by the encouraging finding, Zhu’s group proposed a 365-nm light initiated topochemical strategy by polymerization of a low molar-mass organic gelator based on a diphenyldiacetylene (DPDA) derivative containing amide groups in para-position. It takes advantage of the incorporation of cholesterol and (R)-α-lipoic acid to realize the absorption regulation and a chiral organogel formation both. In-situ topochemical reaction initiated by 365-nm light can be efficiently conducted to afford polymeric species, whereby the three-dimensional hydrogen bond network plays a key role to facilitate the photopolymerization process. Furthermore, the process can be facilely indicated by distinctive photoluminescent enhancement along with gel transformation on the basis of enlarged π-conjugated skeleton of DPDA, providing a valuable methodology for latent application as fluorogenic materials.
This work has been published on the journal of Chemistry – An Asian Journal with a PhD’s degree candidate Mingjie Zhu as the first author and Professor Liangliang Zhu as the corresponding author. DOI: 10.1002/asia.202100468.
Full text link: https://doi.org/10.1002/asia.202100468.
Get to know us better now!

Wechat:FDUMMers
Search!
Search across our website
Revenant @ 2018 by fudan | All Rights Reserved
Powered by Weicheng

