 +565 975 658
+565 975 658
 info@premiumcoding.com
info@premiumcoding.com
 Monday - Friday, 8.00 - 20.00
Monday - Friday, 8.00 - 20.00
In spite of many publications of chemotherapy and immunotherapy etc., the first choice of clinical treatments of most solid tumors is still surgery resection, and then postoperative cancer recurrence and metastasis get to be the main cause of mortality. On the one hand, the residual tumor cells afford, unfortunately, the seeds for recurrence and metastasis. On the other hand, surgical resection might result in the immunosuppressive microenvironment. Therefore, to achieve optimal oncological control leads to a large resection frequently, which means a part of normal tissue is inevitably resected during the surgery. The guidelines for surgeons have been indicating that leaving tumor cells at the cut edge of the surgical cavity is unacceptable for surgery treatment albeit accompanied with the postoperative adjuvant therapy. Hence, it is of great significance that a surgical treatment can achieve an optimal surgical margin to conserve normal tissues, or even an incomplete resection of the malignancy with a positive surgical margin does not matter.
The emergence of cancer immunotherapy in recent years has opened a new chapter with the birth of cancer vaccines, adjuvants, adoptive cell therapy, and checkpoint blockade, etc. In general, vaccines can be divided into two classes: prophylactic vaccine and therapeutic vaccine. Recently, Ding’s group at Fudan University has introduced a therapeutic-prophylactic vaccine (TPV) for the first time. The TPV-gel could be employed as a postoperative filler gel, encapsulating calcium salts and adjuvant R837 to achieve immunogenic cell death and promote immune responses, which eradicates residue tumor (therapeutic) and prevents recurrence and metastasis (prophylactic).
The principle of such a TPV-gel is presented in Figure 1.
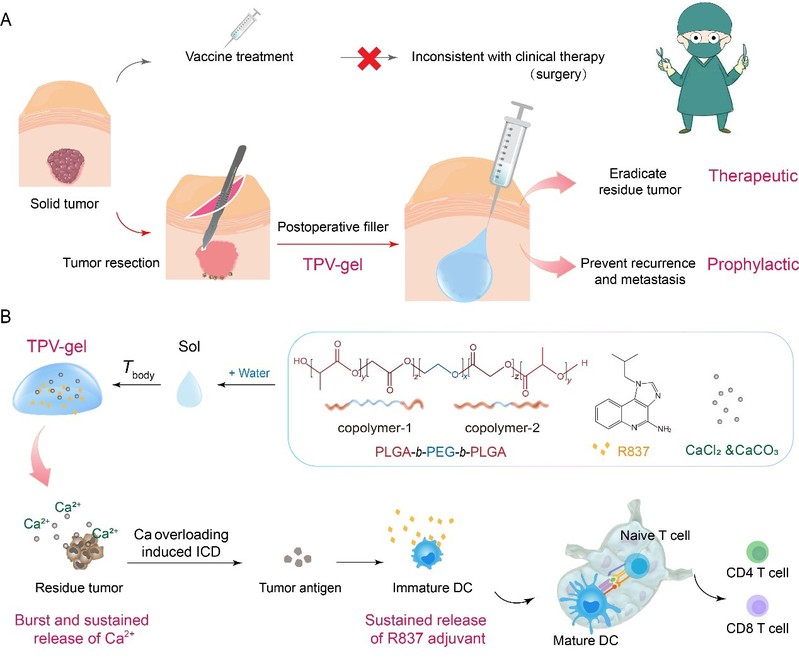
Figure 1. Schematic presentation of the basic idea of the therapeutic-prophylactic vaccine (TPV).
The TPV matrix is initially a polymeric aqueous system composed of PEG/PLGA block copolymers (Figure 2). Considering that the thermogellable composition window for a one-component copolymer is very narrow, Ding’s group put forward a “block blend” strategy. One of the amphiphilic copolymers is relatively hydrophilic (copolymer-1), the other is relatively hydrophobic (copolymer-2). Each of them cannot exhibit sol-gel transition in water with temperature changing, but the appropriate blends can; the sol-gel transition temperature can be well tuned as well. The TPV physically gels at body temperature.
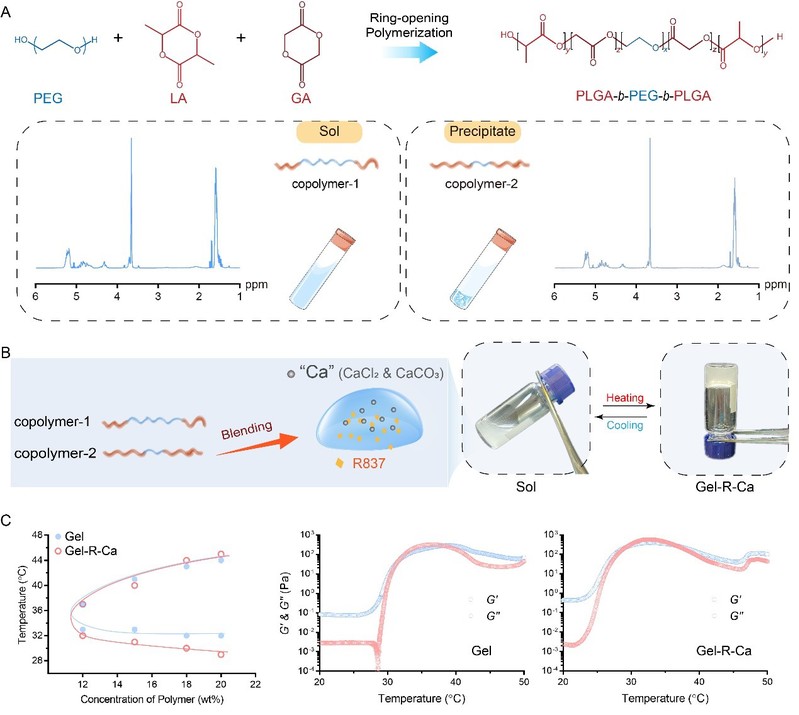
Figure 2. The preparation and gelation of TPV-gel.
The TPV-gel was encapsulated with both calcium-containing inorganic salts (CaCl2 and CaCO3) and the R837 immunoadjuvant (Figure 3). The TPV-gel could achieve a therapeutic effect by providing sufficient calcium ions in residue tumor site and induce immunogenic cell death of residue tumor cells via calcium overload. Appropriate release profiles were achieved after the thermogel was modulated via molecular engineering of polymers.
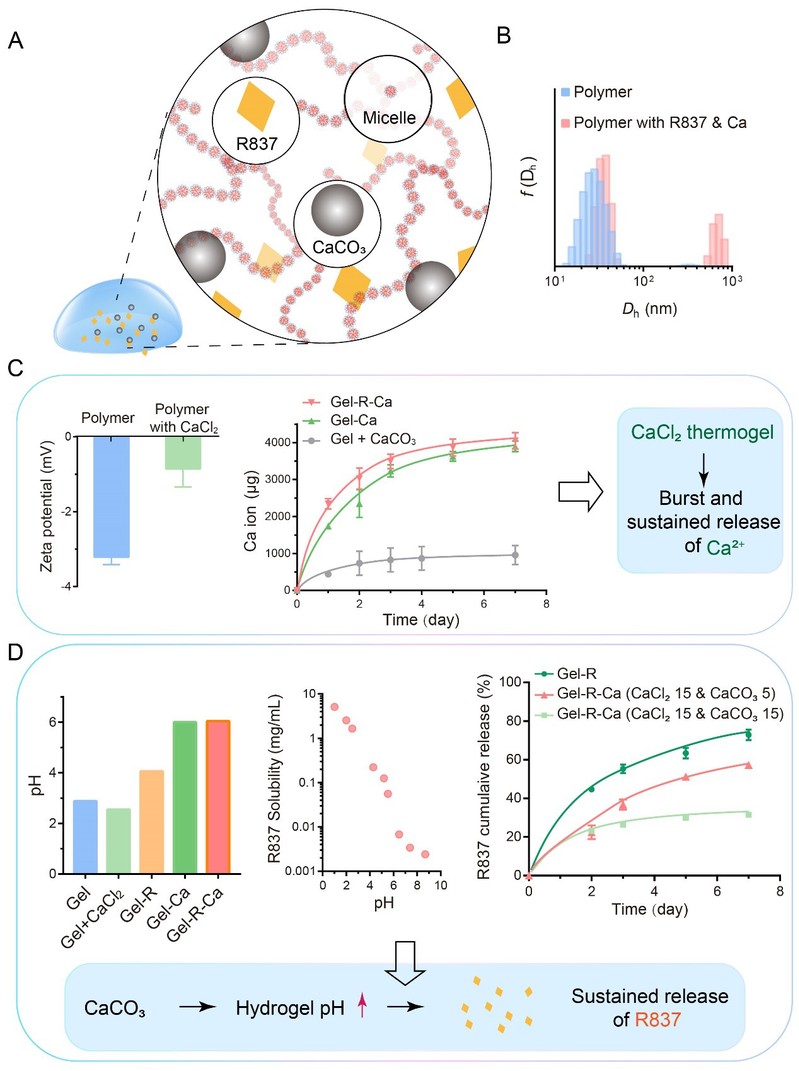
Figure 3. The release profiles of the TPV-gel.
Some in vivo data are shown in Figure 4. The results confirmed the effectiveness of the TVP-gel.

Figure 4. In vivo experiment to demonstrate the efficacy of the TPV-gel.
This work was published in Advanced Functional Materials with a PhD candidate Ms. Dinglingge CAO as the first author, and her supervisor Prof. Jiandong DING as the corresponding author. See please: Cao, D., Guo, W., Cai, C., Tang, J., Rao, W., Wang, Y., Wang, Y., Yu, L., Ding, J.*, Unified Therapeutic-Prophylactic Vaccine Demonstrated with a Postoperative Filler Gel to Prevent Tumor Recurrence and Metastasis. Adv. Funct. Mater. 2022, 2206084.
Article link: https://doi.org/10.1002/adfm.202206084
Get to know us better now!

Wechat:FDUMMers
Search!
Search across our website
Revenant @ 2018 by fudan | All Rights Reserved
Powered by Weicheng

